Anesthesiologists recommend Covalon’s gentle and effective silicone solution over acrylic dressings for securing vascular insertion sites in Epidermolysis Bullosa patients prone to skin injury
Covalon Technologies Ltd. (the "Company" or "Covalon") (TSXV: COV; OTCQX: CVALF), an advanced medical technologies company, today announced that its IV Clear® line of dual antimicrobial silicone dressings has been recommended by physicians at a Top 10 U.S. Children’s Hospital in Colorado, which is also an EB (“Epidermolysis Bullosa”) Center of Excellence, as a novel solution to effectively secure and protect vascular access insertion sites on patients with fragile skin, including those with EB.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20230613612362/en/
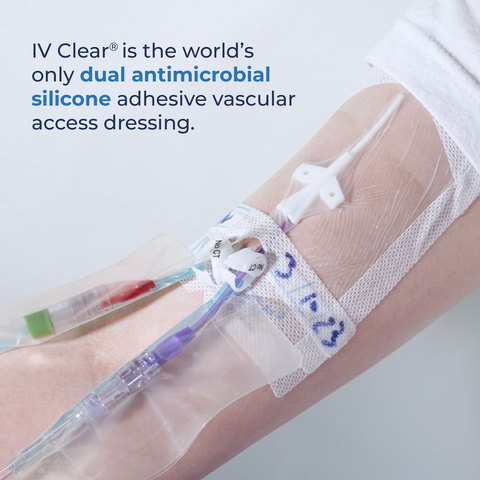
Covalon's IV Clear® is the world's only dual antimicrobial silicone adhesive vascular access dressing. (Photo: Business Wire)
EB, sometimes known as “Butterfly Skin”, is a little-known skin disorder with a prevalence of 1 in 20,000 births. There is no cure to the debilitating connective tissue disorder that causes extremely fragile skin prone to blisters and tears from minor friction. EB patients often require surgical procedures, and securement of intravenous (IV) catheters and peripheral nerve catheters on EB patients present major challenges to anesthesiologists.
In a statement, Kim Strupp MD, FAAP and Norah Janosy, MD, said, “As pediatric anesthesiology physicians who care for patients with Epidermolysis Bullosa (EB) routinely when they need surgical interventions or procedural care, IV Clear® has been transformative in our practice. We are now able to place this transparent, occlusive dressing over intravenous, central and nerve block catheters without damaging our EB patients’ fragile skin. Having a clear, transparent, occlusive dressing decreases the risk of infection, increases the visibility of the catheter, and has improved our care and patient and family satisfaction.”
The recommendation follows research completed by physicians at Children’s Hospital of Colorado (“CHCO”), where they observed how the use of IV Clear® in EB patients improved the safety of indwelling catheter placement and securement without causing injury. The findings were presented by Drs. Mancone, Strupp, Janosy, and Brooks Peterson at the International Anesthesia Research Society meeting in Denver, Colorado on April 14, 2023.1 To date, IV Clear® dressings have been used on approximately 20 EB patients at CHCO without incidence of skin damage or other adverse events. One patient who spent several months in the hospital underwent several consecutive dressing applications without incident.
“At Covalon, our mission has been driven by compassion and a commitment to improving patient care,” said Ron Hebert, SVP Marketing of Covalon. “Healing should never be hindered by setbacks, trauma, or tears caused by dressings. IV Clear® is designed to ensure that every step of a patient’s journey is one of comfort and compassion.”
Traditional securement dressings made with acrylic cause substantial injury upon removal. Covalon’s IV Clear® contains atraumatic silicone adhesive and its gentle, dual antimicrobial properties make it ideal for use on patients at risk of skin injury.
Covalon’s vascular access infection prevention solutions also help to prevent central line-associated bloodstream infections (CLABSIs) as part of an effective CLABSI prevention bundle, while ensuring patient comfort and include:
- CovaClear® IV – utilizes soft silicone adhesive technology to help protect patients from skin injuries, but does not incorporate antimicrobials, for use with patients who either don't require, or cannot tolerate, antimicrobials.
- IV Clear® - the world’s only dual-antimicrobial vascular access dressing that offers complete transparency at and around the insertion site for easy daily assessment. It also utilizes soft silicone adhesive technology to minimize skin injuries and preserve skin barrier functions, and incorporates safe amounts of antimicrobials, without sacrificing efficacy, to protect against chemical irritation.
- VALGuard® - an FDA listed, transparent, environmental barrier designed to protect catheter hubs and line connections from external contaminants and gross contamination, including body fluids and other secretions. It incorporates a quick-release pull strip for fast access to infusion hubs and for easy removal.
Those interested in learning more about Covalon’s solutions may visit www.covalon.com or follow Covalon on LinkedIn, Facebook, Instagram or Twitter.
Reference
- Mancone, A. et al. 2023. Novel Approach to Catheter Securement for a patient with Epidermolysis Bullosa. [Poster]. IARS Annual Meeting 2023, 14 April, Denver.
About Covalon
Covalon Technologies Ltd. is a patient-driven medical device company, built on the relentless pursuit to help the most vulnerable patients have a better chance at healing. Through a strong portfolio of patented technologies and solutions for advanced wound care, infection prevention, and medical device coatings, we offer innovative, gentler, and more compassionate options for patients to heal with less infections, less pain, and better outcomes. Our solutions are designed for patients and made for care providers. Covalon leverages its patented medical technology platforms and expertise in two ways: (i) by developing products that are sold under Covalon’s name; and (ii) by developing and commercializing medical products for other medical companies under development and license contracts. The Company is listed on the TSX Venture Exchange, having the symbol COV and trades on the OTCQX Market under the symbol CVALF. To learn more about Covalon, visit our website at www.covalon.com.
Anesthesiologists at a Top 10 U.S. Children’s Hospital in Colorado, which is also an Epidermolysis Bullosa Center of Excellence, recommend Covalon’s gentle and effective silicone solution over acrylic dressings for securing vascular insertion sites in EB patients prone to skin injury.
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
This news release may contain forward-looking statements which reflect the Company's current expectations regarding future events. The forward-looking statements are often, but not always, identified by the use of words such as "seek", "anticipate", "plan, "estimate", "expect", "intend", or variations of such words and phrases or state that certain actions, events, or results “may”, “could”, “would”, “might”, “will” or “will be taken”, “occur”, or “be achieved”. In addition, any statements that refer to expectations, projections or other characterizations of future events or circumstances contain forward-looking information. Statements containing forward-looking information are not historical facts, but instead represent management’s expectations, estimates, and projections regarding future events. Forward-looking statements involve risks and uncertainties, including, but not limited to, the factors described in greater detail in the “Risks and Uncertainties” section of our management’s discussion and analysis of financial condition and results of operations for the year ended September 30, 2022, which is available on the Company’s profile at www.sedar.com, any of which could cause results, performance, or achievements to differ materially from the results discussed or implied in the forward-looking statements. Investors should not place undue reliance on any forward-looking statements. The forward-looking statements contained in this news release are made as of the date of this news release, and the Company assumes no obligation to update or alter any forward-looking statements, whether as a result of new information, further events, or otherwise, except as required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20230613612362/en/
“As pediatric anesthesiology physicians who care for patients with Epidermolysis Bullosa (EB) routinely when they need surgical interventions or procedural care, IV Clear® has been transformative in our practice."
Contacts
To learn more about Covalon:
Brian Pedlar, CEO, Covalon Technologies Ltd.
Email: bpedlar@covalon.com
Phone: 905.568.8400 x 233
Toll-Free: 1.877.711.6055
Website: https://covalon.com/
Twitter: @covalon












